In-house cytology gives dermatology patients more bang for their buck
Dermatology requires a methodical, step wise approach and begins with ruling out secondary infections
Photos courtesy Jennie Tait
In-house cytology is by far the single best thing you can do for your dermatology patients. The skin is a uniquely accessible organ, which allows for high yield sampling to be easily performed. For dermatology cases, in-house cytology can provide accurate diagnoses within minutes at a very low cost. Affordable and efficient patient care? Talk about a win-win!
Dermatology cases are usually convoluted. Indeed, the vast majority of these patients have secondary infections that exacerbate primary conditions. After working for 22 years in a dermatology referral practice, this author can attest that performing cytology in these cases is an absolute must—this goes for patients getting an initial assessment, as well as those visiting the clinic for re-checks to monitor response to therapy.
Even with my extensive experience in the field, I still find myself surprised by cytologic findings after analysis. There is simply no way to know for certain what kind of infection (bacterial or fungal) you are dealing with until samples have been assessed under the microscope.
Sample collection
There are several ways one can collect samples for evaluation, as well as varied reasons for implementing different techniques.
Collection usually takes the form of direct smears or acetate tape samples. Direct techniques include impressions, swabs, and scrapings. All of these involve taking superficial samples from the surface of the skin.
Impressions
Impressions are collected by pressing a slide to the skin several times. This method of collection works best in cases of greasy or moist lesions. It can also be used when lancing an intact pustule with a sterile needle or lifting a crust to reveal an exudative lesion. A lifted crust can also serve as a stamp on the slide to provide an impression smear. Impression smears are the preferred method when looking for acantholytic keratinocytes associated with pemphigus.
Swabs
Swabbing is done using a cotton-tipped applicator, which is rubbed vigorously (like colouring with a crayon) on a lesion, then gently rolled out as a stripe on a microscope slide.
To save time and money, consider rolling out multiple samples from different lesions on the same slide—this means there is only one slide to process and analyze. When doing this, I start on the left (closest to the frosted edge) with my first sample, writing the site on the frosted end of my slide with a pencil, then add samples working across to the right (Figure 1). When working on ears, I put the left-ear sample on the left side and the right-ear sample on the right. (To make this extra idiot-proof, I roll out an ‘L’ with my left-ear sample.)

Scraping
Scraping collects surface debris from the skin using either a dulled scalpel blade or a clean spatula. The collected debris is smeared on to a microscope slide. Scrapings for cytology are superficial and use no oil.
While collecting samples is pretty straightforward, it helps to have a gentle hand. This is nicer for the patient, and also avoids what I like to call ‘schmooshocytes’—or, nuclear streaming, whereby cells (usually neutrophils) are ‘schmooshed,’ smearing their nuclear contents. This unfortunate scenario makes interpreting findings extra challenging. The damage happens most often with direct swabs, as well as overzealous impression smears. These are also called ‘touch’ preps (not ‘push-so-hard-you-break-the-slide’ preps!). If you have ever collected samples on a farm call only to find schmooshocytes upon evaluation back at the clinic, you know how disappointing this can be.
Staining slides
There are many ways to stain smears. Here are some helpful tips and steps to keep in mind:
1) Samples from dry, waxy, or greasy (not moist) skin should be heat-fixed, as this keeps the sample in place during processing. After heat-fixing, wipe the back of the slide to remove any possible soot left behind. All direct techniques should be stained with Diff-Quik (fixative + two stains, rinse, dry, coverslip) and examined under a microscope for evaluation. Samples from moist lesions or FNAs should be air dried prior to staining.
2) Dip the slide in fixative and gently wobble it back and forth for a slow count of 10, then remove it from the fixative and allow any excess to drain into fixative container. Dip the slide into pink eosin stain and gently wobble back and forth for a slow count of 10. Remove the slide and allow any excess to drain into the stain container.
3) Dip the slide into the final purple, basophilic stain and gently wobble it back and forth for a slow count of 20. Doubling the time of the purple stain allows for better saturation of things like yeast, mast cells, and acantholytic keratinocytes.
4) Remove the slide from the stain and allow any excess to drain into the purple stain container. Rinse both sides of the slide with a gentle stream of tap water. When you think you are done, give it one more rinse. Runoff from the slide should be clear.
5) Blot the slide dry using a clean section of bibulous paper. Do not rub the sample; simply press down. Apply a drop of immersion oil followed by a coverslip and the sample is ready to be evaluated. (Note: using a long [22-mm x 40-mm] coverslip allows for a larger area to be easily read, which is handy if more than one sample is rolled onto the slide.)
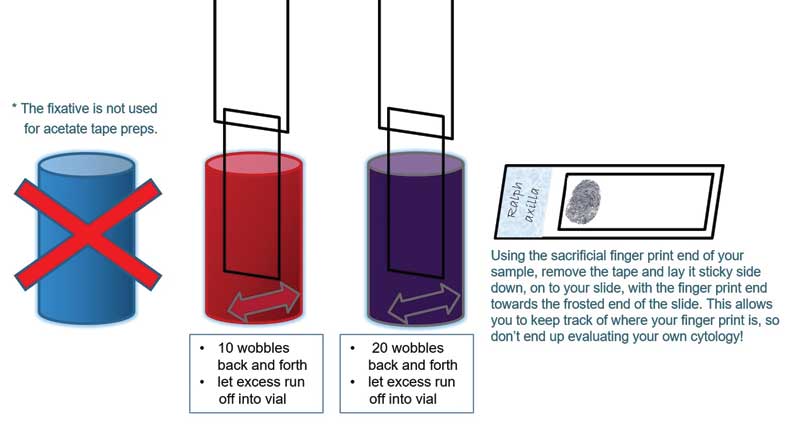
Tape
There are countless ways to process tape samples. My tried-and-tested technique (Figure 2) has evolved over the years to one that is quick and high yield—this means I can find parasites, fungal spores and hyphae, pollens, inflammatory cells, bacteria, yeast, and skin cells all in one go.
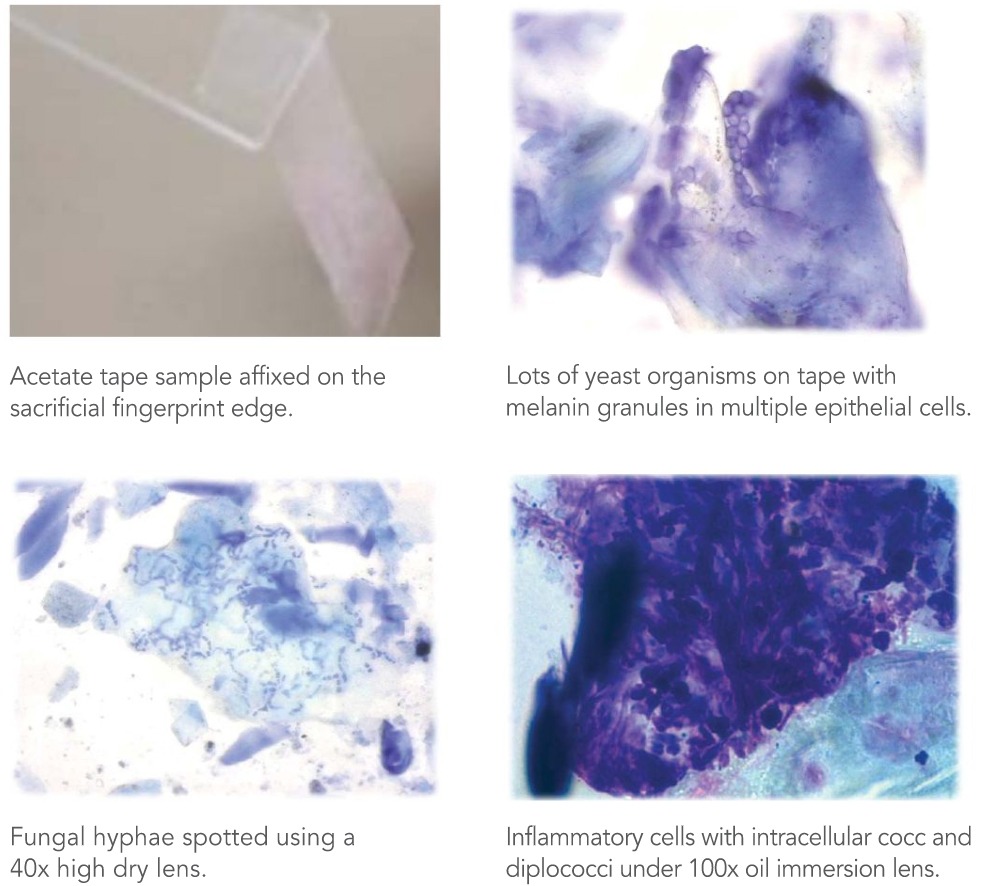 1) Start with clear acetate tape.
1) Start with clear acetate tape.
2) Handle the tape strip by one end only—this is your ‘sacrificial fingerprint end.’ Keep the tape edge stuck to your finger, apply the sticky surface to the area for collection, and press down using your thumb. Re-apply to the site at least three times.
3) Handle only the fingerprint end and stick this edge to the clear end of a microscope slide. Press to be certain it is well attached. (Alternatively, you may consider handling the fingerprint end of tape with forceps for the staining sequence.)
4) Do not (I repeat—do not!) use the fixative in your stain set! This will dissolve the glue on the tape, taking your sample with it.
5) Dip the free end of the tape into the pink, eosin stain and wobble it back and forth for a slow count of 10. Remove and allow any excess stain to drip back into the container.
6) Dip the free end of the tape into the final, purple stain and wobble it back and forth for a slow count of 20. Fungal organisms, including yeast, can take longer to stain, so give it time. Remove and allow any excess to drip back into the container.
7) Rinse both sides of the tape with copious amounts of water (I use tap water—I have experimented with distilled and deionized water for years, but have found it does not make any discernable difference). When you think you are done, give each side one more rinse. Your runoff should be clear.
8) Remove the tape from the slide’s edge and apply it, sticky side-down, to the length of the microscope slide with your sacrificial fingerprint end nearest to the frosted end of your slide. This prevents you from discovering what is on your own skin surface instead of that of your patient.
9) Lay the slide on a paper towel. Fold the towel over and press, moving from one end of the slide completely across to the other side. This removes any air bubbles and excess fluid. Repeat this a second time.
10) To ensure the clearest image possible, apply a drop of immersion oil, followed by a long coverslip. Tape samples take some time to get used to—they are very busy (i.e. extremely high yield), and things look a little different, especially neutrophils. You tend to see only the nucleus of neutrophils, so I look for ‘fuchsia squiggles’ under low power, then investigate those areas further under high power. If present, intracellular and extracellular bacteria can be spotted (along with all of the other usual suspects).
Generally, examine cytology samples under the lowest power lens first, then gradually move up through higher objective lenses and finish with examining under oil immersion. This final step ensures no small organisms are missed. Be sure to use your fine focus to tease out anything that could be hiding on different planes.
Personally, I am a huge fan of tape preps (ask any of my colleagues!); however, I will not reach for the tape if a lesion is very moist. In these scenarios, it is always preferable to, instead, opt for an impression smear or a swab.
This begs a follow-up question: how does one know when to use which technique, anyway? While this largely comes down to preference, there are certain scenarios when one method offers greater advantage over another (Table 1).
Final thoughts
When it comes to best practices for in-house cytology, it is essential everyone in the clinic ‘speaks the same language.’ This ensures medical reports mean the same thing to everyone, which is especially important for gauging response to treatment. To help with this, the Canadian Academy of Veterinary Dermatology (CAVD) offers many printable resources on its website (cavd.ca). Posting guides such as these next to the microscope can help everyone quantify what, exactly, 1+, 2+, 3+, and 4+ mean.
Additionally, for best success with your in-house cytology, be sure to minimize any artifacts as much as possible—this means copious rinsing of stained samples and changing stains at least once a week.
Dermatology is far from rocket science. Much like any other case in the clinic, it requires a methodical, step wise approach and begins with ruling out secondary infections. Performing simple, low-cost, in-house cytology can help ensure patients are receiving an accurate diagnosis and appropriate treatment protocols in the timeliest of manners.
Jennie Tait, AHT, RVT, VTS (Dermatology – Charter Member), is a member of the health care team at the Veterinary Allergy Dermatology Ear Referral (VADER) Clinic in Morriston, Ont. She has served as a regent for the Academy of Dermatology Veterinary Technicians since its inception in 2009 and is on the executive committee for the Canadian Academy of Veterinary Dermatology (CAVD). Tait has nearly 25 years of experience teaching veterinary students at the Ontario Veterinary College and has 22 years’ experience specializing in veterinary dermatology. Currently the only VTS (Dermatology) in Canada, she graduated as an Animal Health Technician from Centralia College of Agricultural Technology in 1986. She is the proud owner of RVT certificate #4 and is an accomplished speaker and author in her area of expertise.




