What’s new in reptile analgesia

There have been substantial advances in the field of reptile analgesia over the last decade, which is critically important for the welfare of these species. However, due to the extreme diversity and numbers (more than 7000 species) within the Class Reptilia, there are still significant gaps in knowledge.
This article will review multiple recent analgesic studies in reptiles, including which drugs are currently recommended and which should be avoided due to their lack of efficacy. It is important to note that most of the referenced studies were performed in healthy reptiles, and the results may be different in a compromised or ill animal.
Pain recognition
Most veterinarians are in agreement reptiles can feel pain, but difficulty recognizing pain in reptiles was cited as a major impediment for providing appropriate analgesia by a recent survey of veterinarians.1 Only ~33 per cent of veterinarians in that survey believed they had adequate knowledge of analgesia in reptiles, but 82 per cent of respondents reported providing analgesia to their reptile patients.1
In another published survey, 76 per cent of reptile veterinarians stated they diagnosed pain in reptiles based on extrapolation from other species, and 66 per cent reported they diagnosed pain from “behavioural changes.”2
While grimace scores and other objective measures of pain have become more commonplace for many mammalian pet species, no such scores exist for any reptile species to date.
Reptiles have limited muscles of facial expression compared with most mammals, and most species do not vocalize. Therefore, objective measurements of pain are difficult to evaluate in most reptile species.
According to an informal survey of reptile veterinarians, the most commonly reported pain behaviours across reptile families included decreased interaction with the environment, decreased appetite, decreased movement, and inability to bear weight on an affected limb.3
The anatomic diversity amongst reptiles also poses a challenge for pain assessment, including the lack of limbs in snakes and some lizard species, and the presence of a shell (plastron and carapace) in chelonians.
The term “pain” is often used to imply a higher level of neural processing, encompassing both a physical and emotional response, such as what occurs in the neocortex of mammals. However, since both birds and reptiles lack a neocortex, the terms nociception and antinociception are likely more appropriate to use to describe “pain” and “analgesia” in most non-mammalian species.4 However, many veterinarians and publications still use these terms interchangeably.
The presence of scaled skin and certain species’ proclivity for self-induced thermal burns (such as from a heating element) pose challenges for designing accurate analgesic efficacy research methods, specifically those that utilize thermal stimuli.
Alternative models have been evaluated in reptiles, and another study found that a decrease in feeding behaviour showed promise to assess antinociception in ball pythons.5
For additional information on pain models and comparisons of the pain pathway between mammals and reptiles, the reader is encouraged to consult
a recently published review article on this subject.3
Opioids
Full mu opioids have been proven to be efficacious in numerous reptile species to date. However, based on several previous studies, it is still unclear whether mu opioids are efficacious in any snake species.6,7
Morphine
Morphine provided antinociception in bearded dragons (1 and 5 mg/kg SC) and red-eared sliders (1.5 and 6.5 mg/kg SC), but there was no clear efficacy proven in corn snakes, even when administered at an extremely high dose of 40 mg/kg.8,9,10 Antinociception was also shown in black and white tegus following single intramuscular injections of morphine at 5 and 10 mg/kg.11 Similar to mammals, respiratory depression has also been seen in higher doses of morphine in reptiles, and clinicians should be aware of this possible side effect if morphine is administered on an outpatient basis or when a respiratory rate cannot be monitored.8
Hydromorphone
Hydromorphone is another mu opioid receptor agonist that has been evaluated in several reptile species, with good analgesic efficacy. In red-eared sliders and bearded dragons, hydromorphone was found to provide analgesia at 0.5 mg/kg SC for up to 24 hours.12,13 Respiratory depression was seen at higher doses, similar to morphine, and clinicians should plan appropriately for monitoring respiration.
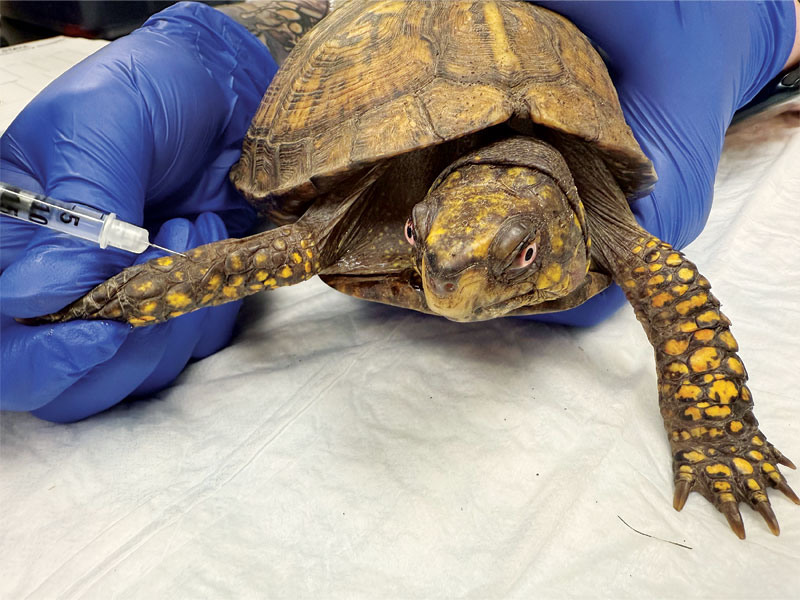
Injections of any drug (analgesic or other) should be performed in the forelimbs (Figure 1) or cranial portions of a reptile’s body, as drugs that undergo hepatic metabolism/excretion or renal tubular excretion can have reduced efficacy and/or enhanced toxicity if injected in the caudal aspects of the body/hindlimbs due to the presence of the renal portal system.
Fentanyl
There are several previous studies that have evaluated fentanyl patches in reptile species. Anecdotally, fentanyl patches have shown promise for providing analgesia in several snake species, particularly for alleviating pain associated with chronic conditions such as spinal osteoarthritis.
As there are limited studies demonstrating any effective analgesics in snakes, this was a promising finding. A 2017 study found that ball pythons had similar concentrations of mu-opioid receptors in their brain and spinal cord tissues as turtles and showed respiratory depression after placement of transdermal fentanyl patches, suggesting the physiologic efficacy and route of this drug.14
In addition, plasma concentrations were high following transdermal administration, suggesting positive transdermal absorption. However, there were no differences seen in response to a thermal stimulus, suggesting a lack of analgesia, at least according to this pain model.14
A subsequent study has recently been published by this same research group, evaluating serum fentanyl concentrations and behaviours after the placement of transdermal fentanyl patches in healthy corn snakes.15 The plasma concentrations of fentanyl in those snakes remained above therapeutic concentrations in mammals for approximately four weeks. In addition, the behavioural changes suggested this dose provided analgesia without negative side effects.15
Buprenorphine
Buprenorphine, a partial mu opioid receptor agonist and kappa receptor antagonist, is frequently used for analgesia in mammals. However, according to studies in green iguanas16 and red-eared slider turtles,17 effective analgesia has not been demonstrated in reptiles. Therefore, this drug is not currently recommended for analgesia in any reptile species until proven otherwise by additional research.
Butorphanol
Butorphanol is a kappa receptor agonist/mu receptor antagonist that is commonly used in many avian species for analgesia and was previously used in reptiles for the same purpose. According to several publications, this drug does not provide analgesia in bearded dragons, corn snakes, ball pythons, black and white tegus, and red-eared slider turtles at a wide range of dosages.6,7 Therefore, butorphanol is also not currently recommended for analgesia in any reptile species.
NSAIDs
Non-steroidal anti-inflammatory drugs (NSAIDs) are commonly used in many species for analgesia and anti-inflammatory properties, including reptiles. They act primarily by inhibiting cyclooxygenase enzymes (COX), which prevents the production of prostaglandins and several other inflammatory mediators.7
The main COX enzymes are COX-1, which are expressed in most tissues, and COX-2, which are elevated secondary inflammatory processes. Consequently, most NSAIDs that are classified according to their COX selectivity: COX-1, COX-2, or both (non-selective).
In addition, NSAID efficacy is difficult to quantify, as plasma concentrations of these drugs often do not reflect the tissue concentrations at the site of inflammation, which further complicates NSAID pharmacokinetic and pharmacodynamic research.7
Several studies have found increased concentrations of COX-1 enzymes in inflamed tissues of box turtles18 and ball pythons19 compared with COX-2 enzymes, suggesting COX-1 selective NSAIDs may be more appropriate for these reptile species.
Meloxicam
Meloxicam is a primarily COX-2 inhibitor, and widely used in exotic animal medicine, including reptiles. However, there is conflicting evidence of its efficacy in several reptile species, despite numerous pharmacokinetic studies in this Class. Ball pythons administered meloxicam 0.3 mg/kg intramuscularly showed no indications of analgesia for a surgical catheterization procedure.20 Conversely, after administration of meloxicam (0.4 mg/kg IM) to bearded dragons, signs of successful antinociception were noted.21 Intramuscular meloxicam administered at 0.2 mg/kg in red-eared slider turtles reached plasma concentrations sufficient to provide analgesia in mammalian species for ~48 hours, but the oral bioavailability was poor.22
Ketoprofen
Ketoprofen is a non-selective inhibitor of both COX-1 and COX-2 enzymes and has been used with increasing frequency in reptiles. In green iguanas, ketoprofen had a long half-life when administered at 2 mg/kg IV, but was slightly shorter when administered intramuscularly (~8 hours).23
The safety of repeated doses of ketoprofen (2 and 20 mg/kg IM for 14 days) were recently evaluated in bearded dragons.24 No adverse systemic effects were noted on biochemical panels or histopathologic examination post-mortem, but there was severe muscle necrosis present at the injection sites of the 20 mg/kg group.
Alpha-2 agonists
Despite the lack of experimental antinociception for many analgesics in snakes, several recent studies have demonstrated positive effects with the alpha-two agonist dexmedetomidine in ball pythons.
Dexmedetomidine is often used in combination with other sedatives for anesthesia in reptiles, at much higher doses than what is typical in most mammals. However, a recent study evaluated the response of ball pythons to a thermal noxious stimulus after administration of 0.1 – 0.2 mg/kg SC dexmedetomidine and found it did produce antinociception at those doses.25 Excessive sedation was not noted, but respiration was depressed, without apnea.
In a follow-up paper by the same investigators, dexmedetomidine was administered at 0.1 mg/kg SC, and antinociception was confirmed.26 In addition, concurrent administration of doxapram (10 mg/kg) helped mitigate the reduced respirations without changing the thermal antinociception.26
While doxapram is not routinely used concurrently with dexmedetomidine in reptiles in clinical practice, the use of dexmedetomidine as an analgesic is becoming more common, as few other proven analgesic drugs for snakes exist.
Olivia A. Petritz, DVM, DACZM, graduated from Purdue University and then completed several internships and a residency in the field of zoo and exotic animal medicine. Dr. Petritz became a diplomate in the American College of Zoological Medicine in 2013, specializing in zoological companion animals (exotic pets). Petritz started an exotics service at a specialty hospital in Los Angeles, Calif., following her residency, and is currently an associate professor of Avian and Exotic Animal Medicine at North Carolina State University.
References
View references for this article online at veterinarypracticenews.com/reptile-analgesia.



